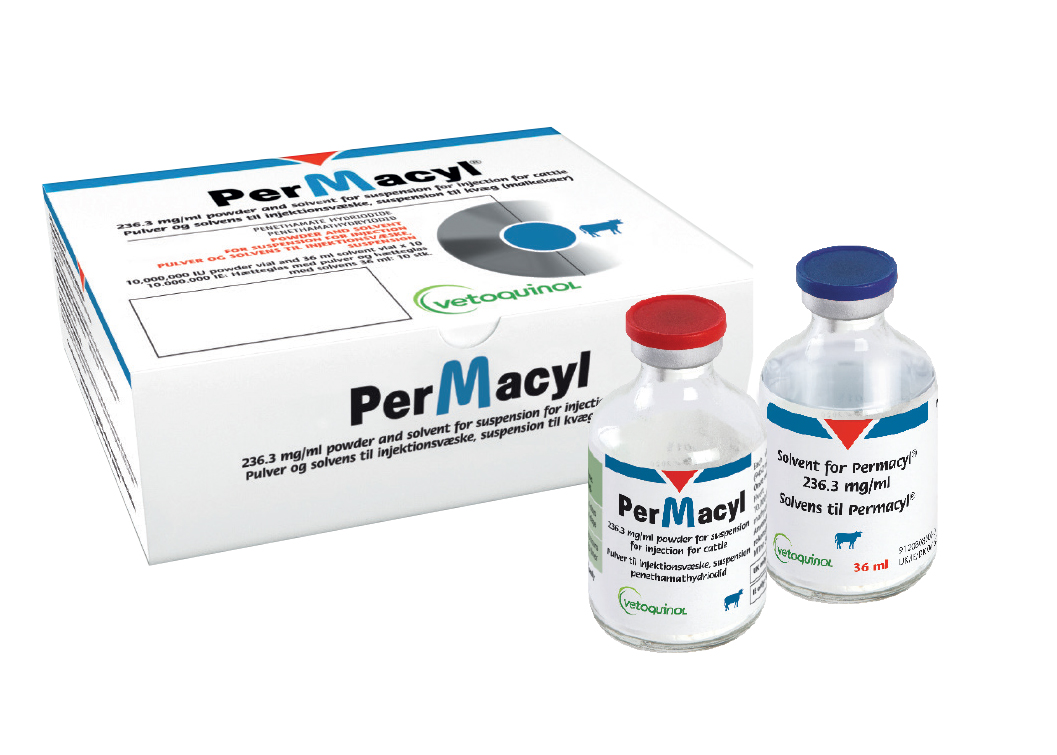
Description
PerMacyl Injection 36ml (pack of 10)
Uses
Treatment of mastitis in lactating cows caused by Streptococcus uberis, Streptococcus dysgalactiae, Streptococcus agalactia and Staphylococcus aureus (beta-lactamase non-producing), sensitive to penicillin.
Dosage and administration
Administer by deep intramuscular injection.
Directions for use: Reconstitute the suspension using the entire contents of the solvent vial.
Shake well after reconstitution. A minimum of 10 inversions of vials can be necessary.
Each ml of suspension contains 250,000 IU (236.3 mg) of penethamate hydroiodide.
Dose: 15,000 IU (14.2 mg) of penethamate hydroiodide per kg of body weight / day (equivalent to 6 ml of reconstituted medicinal product / 100 kg body weight) for three to four consecutive days. Shake well before use.
Administer the recommended daily dose every 24 hours, for three to four consecutive administrations.
To ensure administration of a correct dose, body weight should be determined as accurately as possible.
The recommended maximum volume to be administered at a single injection site is 20 ml.
The stopper should not be punctured more than 10 times.
Contra-indications, warnings, etc.
Contraindications
Do not use in animals known to be hypersensitive to penicillin, cephalosporins, and/or any of the excipients.
Do not administer intravenously.
Do not use in lagomorphs and rodents such as guinea pigs, hamsters or gerbils.
Do not administer to animals with renal disease including anuria or oliguria.
Special warnings for each target species
Treatment should be carried out during lactation.
Special precautions for use
This veterinary medicinal product does not contain any antimicrobial preservative.
Special precautions for use in animals
Using penethamate hydroiodide for the treatment of mastitis must be accompanied by hygienic measures to prevent reinfection.
Where local (regional, farm-level), epidemiological information indicate possible reduced susceptibility of the relevant strains of the bacterial species involved in mastitis, use of the product should be based on susceptibility testing on bacteria isolated from diseased animals.
The veterinary medicinal product is not effective against beta-lactamase producing organisms.
Official national and regional antimicrobial policies should be taken into account when the product is used.
Use of the product deviating from the instructions given in the SPC may increase the prevalence of bacteria resistant to benzylpenicillin and may decrease the effectiveness of treatment with other beta-lactam antimicrobials due to the potential for cross-resistance.
Operator warnings
Penicillin and cephalosporins may cause hypersensitivity (allergy) following injection, inhalation, ingestion or skin contact. Hypersensitivity to penicillin’s may lead to cross-reactions to cephalosporins and vice versa. Allergic reactions to these substances may occasionally be serious.
Do not handle this product if you know you are sensitized, or if you have been advised not to work with such preparations.
Handle this product with great care to avoid exposure.
Wear gloves when handling the veterinary medicinal product to avoid contact sensitization.
In case of accidental self-injection or if you develop symptoms following exposure such as a skin rash, you should seek medical advice and show the package leaflet or the label to the doctor
Swelling of the face, lips or eyes or difficulty with breathing are more serious symptoms and require urgent medical attention.
Wash hands after use.
Adverse reactions (frequency and seriousness)
The symptoms of adverse reactions range from mild skin reactions such as urticaria and dermatitis to severe reactions such as anaphylactic shock with tremors, vomiting, salivation, gastrointestinal disorders and laryngeal edema.
In some situations the treatment may lead to secondary infections due to overgrowth of non-target organisms.
Use during pregnancy, lactation or lay
Can be used during pregnancy and lactation.
Interaction with other medicinal products and other forms of interaction
The product should not be administered with antibiotics that have a bacteriostatic mode of action.
Overdose (symptoms, emergency procedures, antidotes), if necessary
In cases of overdose, adverse reactions such as those described in Adverse Reactions may occur.
Withdrawal periods
Meat and offal: 4 days
Milk: 60 hours
Pharmaceutical precautions
Before reconstitution, the powder and solvent vials do not require any special storage conditions.
The reconstituted suspension should be stored in the refrigerator (2-8°C).
Shelf life after reconstitution according to directions: 24 hours
Legal category
POM-V
Packaging Quantity
Cardboard box containing:
10,000,000 IU powder vial and 36 ml solvent vial
Powder vial: 50 ml type II colorless glass vial closed with a Bromo butyl stopper and sealed with an aluminum flip-top seal. Solvent vial: 50 ml type II colorless glass vial closed with a Bromo butyl stopper and sealed with an aluminum flip-top seal
PerMacyl Injection 36ml (pack of 10)









Reviews
There are no reviews yet.